
[2026/03/10] 近日,IVIEW Therapeutics Inc.创始人兼首席科学家梁波博士收到国际眼科创新联盟(IAOI)的正式邀请,将作为特邀嘉宾出席于2026年3月20日-21日在中国苏州举行的2026 Eyennovation国际峰会。……[获取详情]

[2026/02/13] 【美国宾夕法尼亚州,2026年2月7日】—— IVIEW Therapeutics Inc.(以下简称“IVIEW”),一家专注于眼科创新药物研发的生物制药公司,今日宣布,其用于治疗白内障术后炎症的创新候选药物IVIEW-1701 的临床三期试验申请(IND)已获得美国食品药品监督管理局(FDA)正式许可。这标志着该药物向全球上市迈出了关键性的一步。……[获取详情]

[2026/01/19] [Cranbury, New Jersey, USA, January 16, 2026] — IVIEW Therapeutics Inc. today announced a fruitful participation at JPM Week 2026 in San Francisco, highlighted by strong investor and strategic partner engagement, insightful regulatory dialogue across multiple high-profile forums and one-to-one meetings.During the week, Houman Hemmati, MD, PhD, Board Director and Chief Medical Adviso……[获取详情]
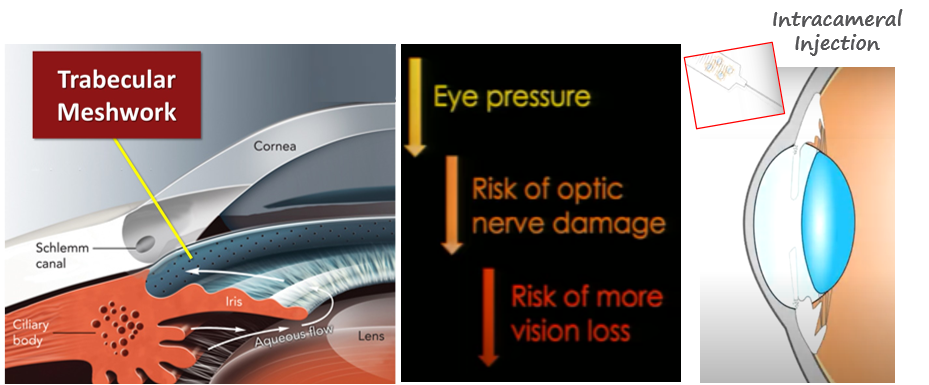
[2025/12/22] 艾威药业(珠海横琴)有限公司(IVIEW Therapeutics 中国子公司)今日正式宣布,其自主研发的创新基因治疗药物 GVB-2001已成功完成原发性开角型青光眼首例患者治疗。该治疗由苏州大学附属第一医院眼科主任陆培荣教授主导实施,目前患者已完成 2 周随访,未发生与药物相关的不良事件,眼压呈现下降趋势,标志着这款适用于所有开角型青光眼患者的基因疗法在人体临床应用中迈出关键一步。……[获取详情]

[2025/12/19] [新泽西州克兰伯里,2025年12月18日]—— IVIEW Therapeutics公司欣然宣布将参与Bio-partnering会议,该会议是 JPM 周系列活动的一部分,是生物技术和生命科学行业最重要的全球性盛会之一。会议汇聚全球生物制药行业领袖、投资者和创新者。……[获取详情]

[2025/11/17] [美国新泽西州,2025年11月13日]– iView Therapeutics(以下简称“艾威药业”)今日宣布,其自主研发的眼科创新药IVIEW-1201D已正式获得美国食品药品监督管理局(FDA)IND 默许同意,开展用于治疗腺病毒结膜炎的关键的III期临床试验。这一里程碑标志着该药物在全球眼科腺病毒结膜炎治疗领域的研发正式迈入全球关键性临床验证阶段,为解决未被满足的临床需求迈出了关键一步。……[获取详情]


 语言:
语言:


